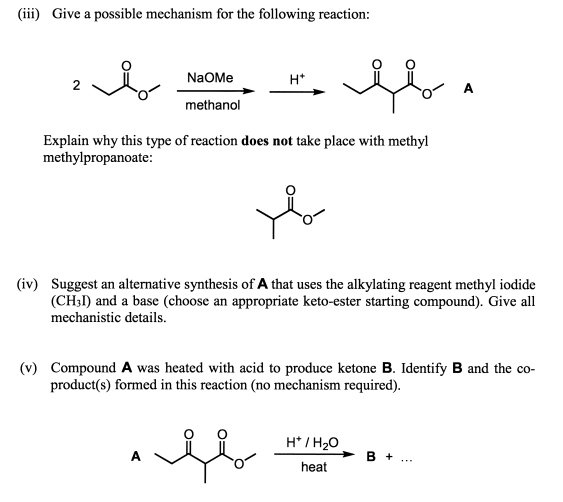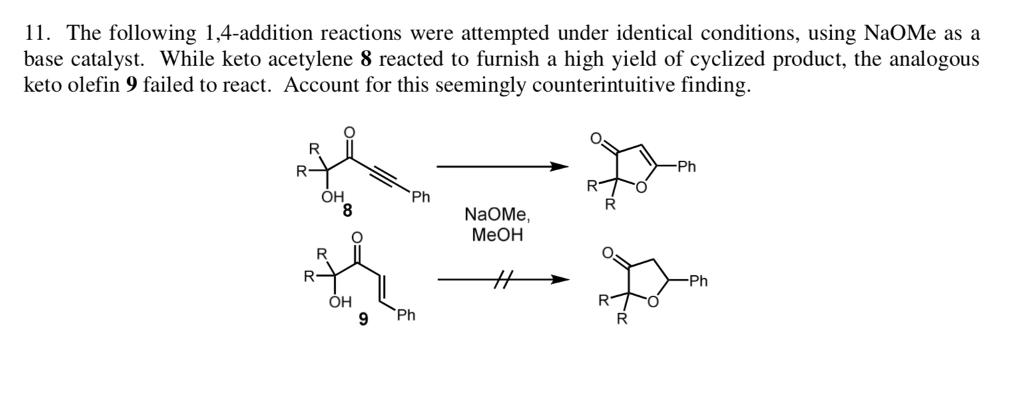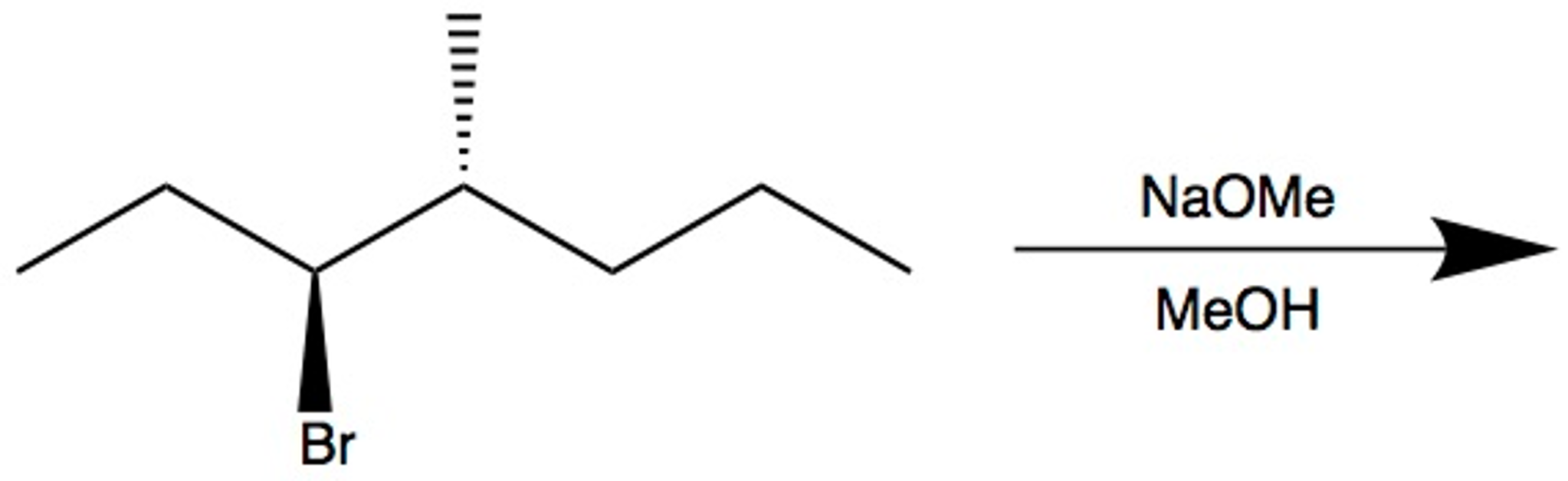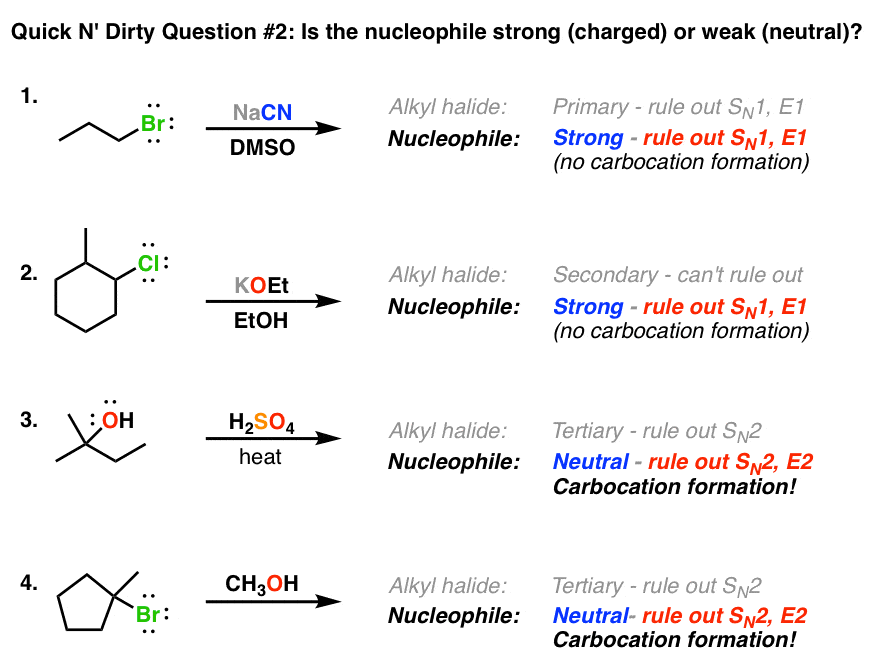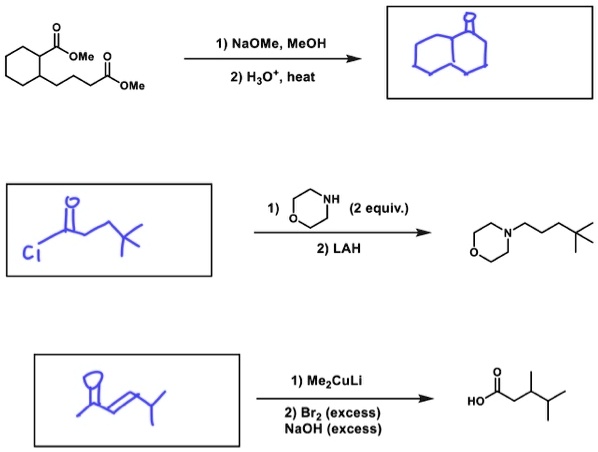
SOLVED: 1) NaOMe, MeOH OMe 2) H3O' , heat OMe (2 equiv:) 2) LAH MezCuLi Brz " (excess) NaOH (excess)

When we use a base in a reaction, why is it always preferred to use the conjugate as the solvent? For example, if NaOMe is my base, the solvent will be HOMe .

Which of the following would be the best base for performing the following elimination? A. NaOH B. NaOMe C. KOtBu D. H2O | Homework.Study.com
organic chemistry - Can the nitrogen of an amide displace a primary chloride in a SN2 reaction in the presence of a strong base? - Chemistry Stack Exchange
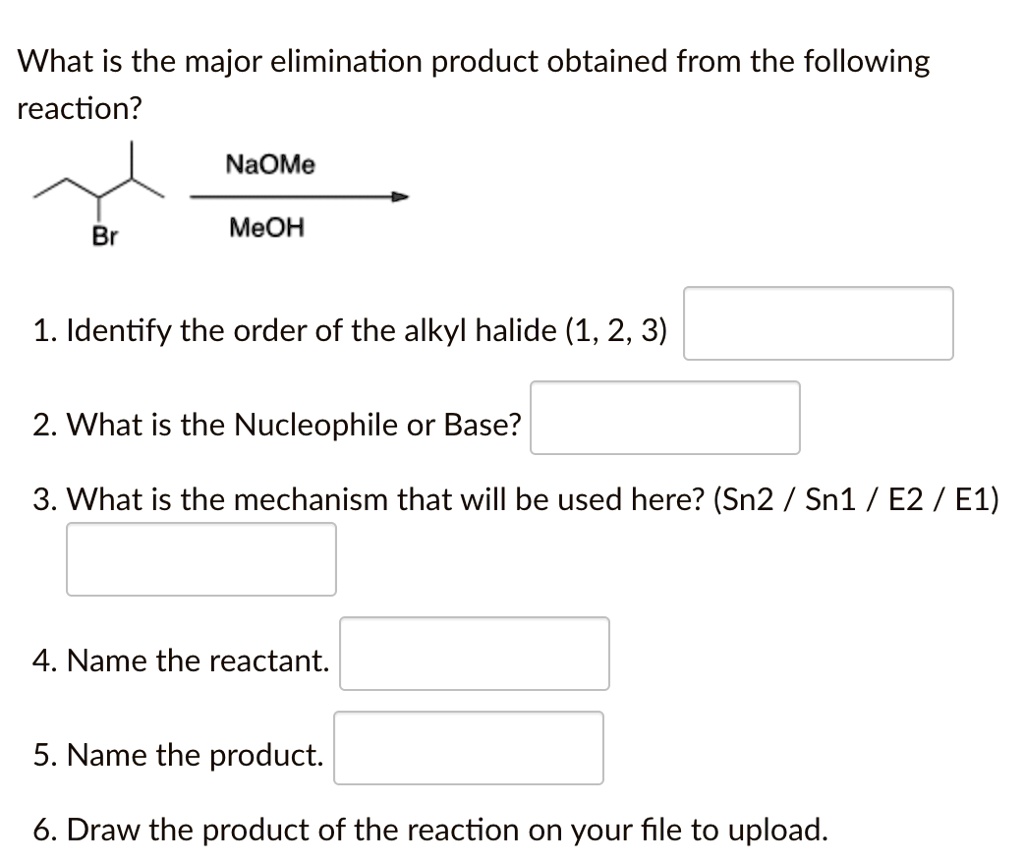
SOLVED: What is the major elimination product obtained from the following reaction? NaOMe Br MeOH 1. Identify the order of the alkyl halide (1,2,3) 2. What is the Nucleophile or Base? 3.
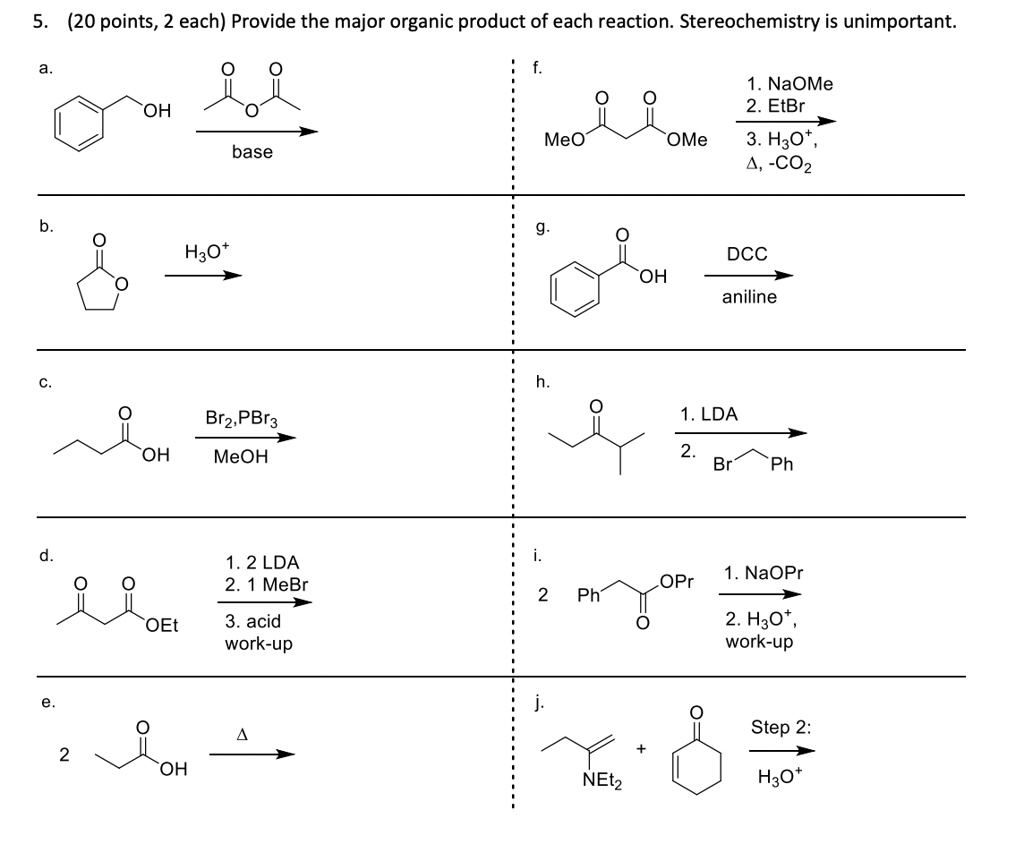
SOLVED: (20 points, 2 each) Provide the major organic product of each reaction. Stereochemistry is unimportant NaOMe 2.EtBr OH Meo OMe 3. H3O COz base Hzot DCC OH aniline 1. LDA OH

When we use a base in a reaction, why is it always preferred to use the conjugate as the solvent? For example, if NaOMe is my base, the solvent will be HOMe .

Scheme 3 Reagents: (a) NaOMe, MeOH; (b) MeI, NaH, THF; (c) TsCl, Et 3... | Download Scientific Diagram

if we had used NaOMe instead of T-BuOK,wouldn't we have ended with the same product? since we have only on Beta postion? : r/chemhelp